Acceleration of 1,3-Dipolar Cycloadditions by Integration of Strain and Electronic Tuning
Jesús M. Dones, Nile S. Abularrage, Namrata Khanal, Brian Gold, and Ronald T. Raines
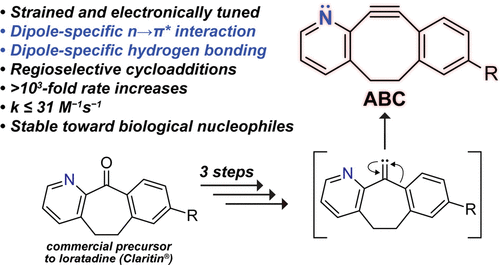 The 1,3-dipolar cycloaddition between azides and alkynes provides new means to probe and control biological processes. A major challenge is to achieve high reaction rates with stable reagents. The optimization of alkynyl reagents has relied on two strategies: increasing strain and tuning electronics. We report on the integration of these strategies. A computational analysis suggested that a CH → N aryl substitution in dibenzocyclooctyne (DIBO) could be beneficial. In transition states, the nitrogen of 2-azabenzo-benzocyclooctyne (ABC) engages in an n→π* interaction with the C=O of α-azidoacetamides and forms a hydrogen bond with the N–H of α-diazoacetamides. These dipole-specific interactions act cooperatively with electronic activation of the strained π-bond to increase reactivity. We found that ABC does indeed react more quickly with α-azidoacetamides and α-diazoacetamides than its constitutional isomer, dibenzoazacyclooctyne (DIBAC). ABC and DIBAC have comparable chemical stability in a biomimetic solution. Both ABC and DIBO are accessible in three steps by the alkylidene carbene-mediated ring expansion of commercial cycloheptanones. Our findings enhance the accessibility and utility of 1,3-dipolar cycloadditions and encourage further innovation.
The 1,3-dipolar cycloaddition between azides and alkynes provides new means to probe and control biological processes. A major challenge is to achieve high reaction rates with stable reagents. The optimization of alkynyl reagents has relied on two strategies: increasing strain and tuning electronics. We report on the integration of these strategies. A computational analysis suggested that a CH → N aryl substitution in dibenzocyclooctyne (DIBO) could be beneficial. In transition states, the nitrogen of 2-azabenzo-benzocyclooctyne (ABC) engages in an n→π* interaction with the C=O of α-azidoacetamides and forms a hydrogen bond with the N–H of α-diazoacetamides. These dipole-specific interactions act cooperatively with electronic activation of the strained π-bond to increase reactivity. We found that ABC does indeed react more quickly with α-azidoacetamides and α-diazoacetamides than its constitutional isomer, dibenzoazacyclooctyne (DIBAC). ABC and DIBAC have comparable chemical stability in a biomimetic solution. Both ABC and DIBO are accessible in three steps by the alkylidene carbene-mediated ring expansion of commercial cycloheptanones. Our findings enhance the accessibility and utility of 1,3-dipolar cycloadditions and encourage further innovation.Available for purchase here.

