News & Events
MSC03 2060
300 Terrace St. NE
Albuquerque, NM 87131-0001
Physical Location:
Clark Hall
Phone: 505-277-6655
chemistry@unm.edu
MSC03 2060
300 Terrace St. NE
Albuquerque, NM 87131-0001
Physical Location:
Clark Hall
Phone: 505-277-6655
chemistry@unm.edu

Profile:
Brian obtained his B.Sc. in chemistry at Florida State University. A short stint in the laboratory of Sir Harry Kroto largely influenced his thinking of science as not simply a journey of personal curiosity, but an avenue to improve society. Continuing his studies at Florida State, he obtained his Ph.D. under the supervision of Prof. Igor V. Alabugin, where he honed his knowledge of organic structure and reactivity through the lens of stereoelectronic effects. He then moved to the frigid tundra of Wisconsin to carry out postdoctoral research in chemical biology, under the supervision of Prof. Ronald T. Raines at the University of Wisconsin–Madison. Another cross-country move—this time with the rest of the Raines Lab—has brought Brian to his current position at the Massachusetts Institute of Technology.
In his relatively short scientific career, Brian has authored nearly 25 publications, including peer-reviewed journal articles, reviews, and encyclopedia chapters. He has (officially) mentored five undergraduate students, including one who continued to obtain her honors thesis. He has also directed or been a part of numerous collaborations in broad research areas.
Abstract:
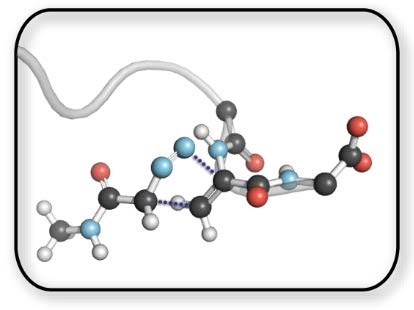 “If one regards a reaction as new only if they have no frontrunners, not even singular examples buried in the literature, then 1,3-dipolar cycloadditions cannot claim novelty. But if one defines reactions as novel which are for the first time recognized for their generality, scope, and mechanism, the judgement must be different.”
“If one regards a reaction as new only if they have no frontrunners, not even singular examples buried in the literature, then 1,3-dipolar cycloadditions cannot claim novelty. But if one defines reactions as novel which are for the first time recognized for their generality, scope, and mechanism, the judgement must be different.”
–Prof. Rolf Huisgen, 1961
The utility of 1,3-dipolar cycloadditions endows this subset of pericyclic reactions with perpetual novelty. The extension of “click chemistry” into the field of chemical biology via the strain-promoted azide–alkyne cycloaddition has provided both tools to understand biology and an arena to showcase the art of controllably manipulating organic reactivity with new standards for selectivity. Without eons of evolutionary optimization, a firm understanding of organic structure and reactivity is our only chance at successfully developing and optimizing ideal bioorthogonal reactions.
See Announcement